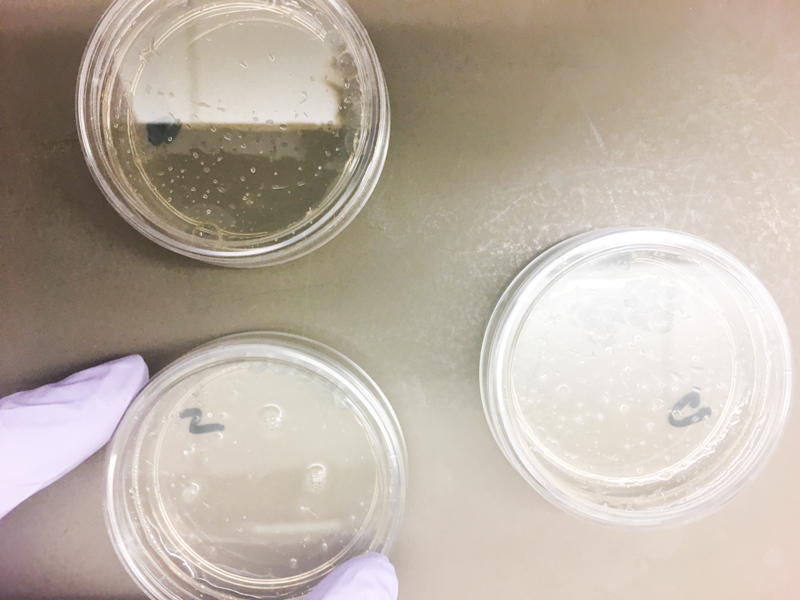
This week we are taking a step towards bringing the cost down of standardized lab equipment. We will be building a low cost electroporator by modifying existing off the shelf hardware. Electroporators cost on the order of $3000, and we will be making one under $10. We will publish more details on the electronic schematics once Teja's project becomes public. We spent most of this week on building the electroporator and calibrating it for proper use. To test our device, we electroporated E.coli cells to open up the cell membrane, and inserted custom pBlu plasmids. For our experiments, we delivered 1,700V during 5ms using the electroporator we fabricated.
We plated the resulting mixture on dishes with LB agar supplemented with ampicillin and X-gal alternatives.
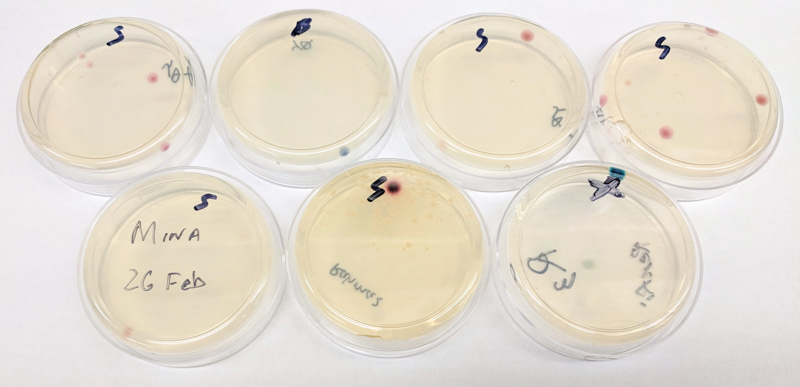
Unfortunately, our cells did not transform as expected, most likely due to a hardware error. One possible hypothesis is that the cells were shocked for too long, or that the waveform is not optimal for opening up the cell membrane. After about four days in the incubator, some cells grew, but the results were not as expected. We will need to tweak the electroporator and try again.
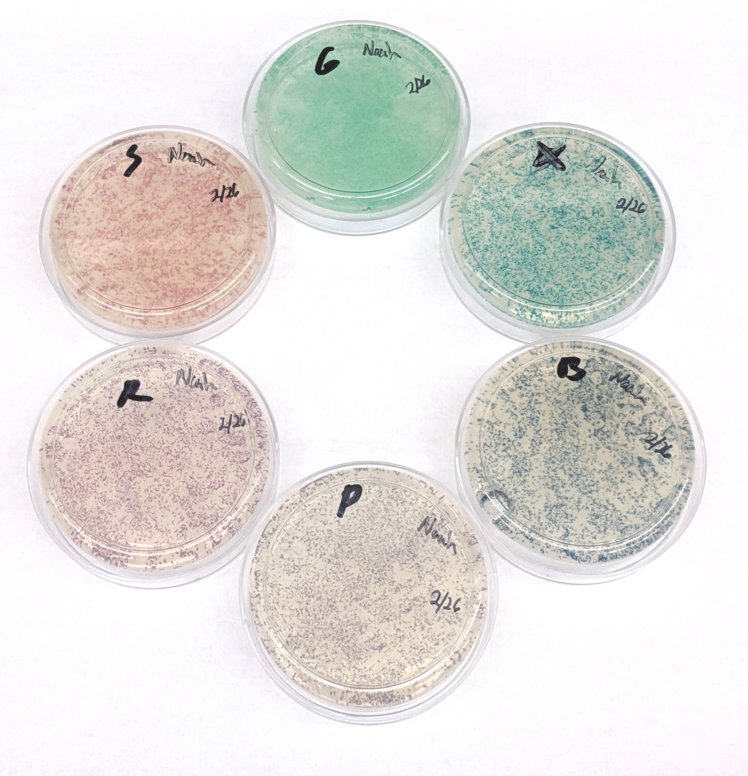
After about four days in the incubator, some cells grew, but the results were not as expected. We will need to tweak the electroporator and try again.In parallel, Noah performed the same experiment but used heat shock to introduce the plasmids into the bacteria instead of heat shock. His result turned out to be much more successful than ours.
One of this week's class highlights was learning about Drew Endy's vision for synthetic biology, where he suggests guidelines for the growth of the discipline based on other engineering fields. The three points he suggests are (i) standardization of components, (ii) component abstraction, (iii) decoupling of design and fabrication.