HTGAA : Next Generation Synthesis
Noah Jakimo (MIT), Joseph Jacobson (MIT)
Assignment
Use synthesized oligonucleotide primers to amplify a PCR product. Assemble a library of protein variants from the PCR amplicons using Gibson Assembly.
Example Lab
We will be changing the color-generating chromophore of the purple Acropora millepora chromoprotein (amilCP) to a variety of orange, pink, and blue mutants. These divergently-colored genetic variants of amilCP were described by Liljeruhm et al in 2018. Their strategy to identify where to mutate amilCP was inferred by sequence similarities to the chromophore region that allows for spectral engineering of the structurally-characterized and well-known green fluorescent protein (GFP), which is native to the jellyfish Aequorea victoria. First we will prepare for a Gibson assembly by using polymerase chain reaction (PCR) to produce two sets of amplicons as inserts and a restriction digest of the common cloning plasmid pUC19 to produce a new backbone (i.e. origin of replication and drug resistance gene). As template, both reactions use the amilCP-encoding plasmid that was miniprepped from the Addgene mUAV sample (deposited by the Nakayama lab at the University of Edinburgh and related to their paper on Mobius Assembly via a Mobius Assembly Universal Acceptor Vector). One set of amplicons copy the region of the amilCP gene that precedes the chromophore, including the transcription promoter and translation ribosome-binding site (RBS). Another set amplicons copy the region that spans 24 basepairs before the chromophore to just beyond the gene's transcription terminators. The latter includes a diversified chromophore-coding segment dictated by mismatches in the PCR primers with respect to the mUAV DNA template. The amplicon sets both include one end that overlaps by 20-22 bases with distinct ends of the large backbone fragment from the pUC19 digest. Lastly, we will express our colorful variety of amilCP mutants in chemically competent E coli cell.
Please review the lecture slides for the assignment.
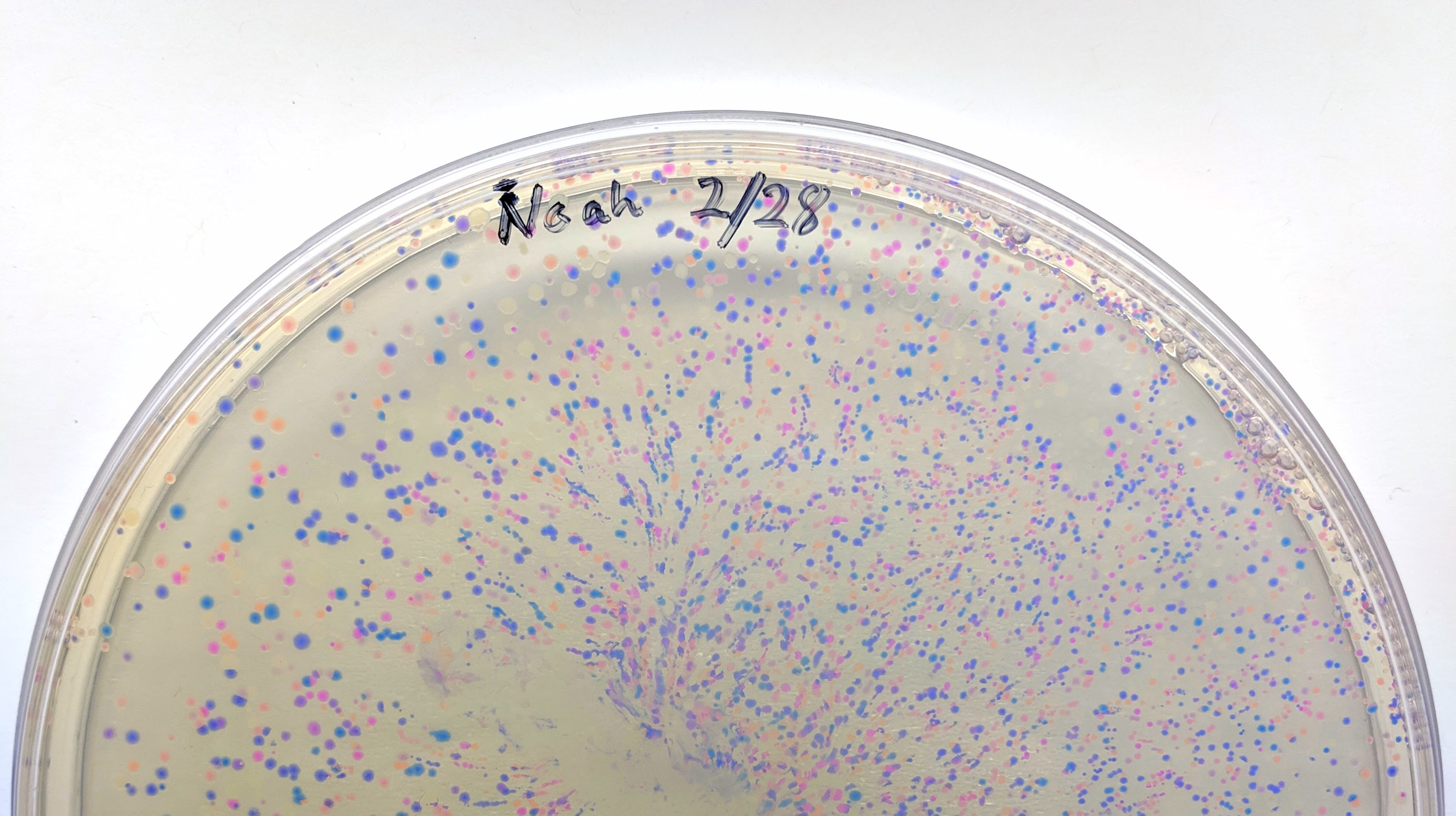
Remember to always initial and label your tubes so you can identify their contents and distinguish them from your classmates' material.
-
Digest the pUC19 plasmid with the restriction enzyme PvuII to generate the linear blunt-ended backbone fragment.
-
Prepare the reaction in a PCR tube (add enzyme last and water first):
To Reaction Volume (50 uL Totral) DNase/RNase-Free Water 40 uL 2 ug pUC19 (1 ug/ul in green cap tubes) 2 uL 10x NEB Buffer CutSmart 5 uL NEB PvuII-HF 3 uL - Incubate reactions at 37C for at least 15 minutes in thermocycler.
-
Prepare the reaction in a PCR tube (add enzyme last and water first):
-
Amplify PCR fragments for the gene assembly of amilCP mutants.
-
Prepare the reaction in a PCR tube to produce the universal chromophore-preceding fragment:
To Reaction Volume (50 uL Totral) DNase/RNase-Free Water 18 uL 200 ng miniprepped amilCP (100 ng/ul in blue cap tube) 2 uL Forward primer with tail that overlaps with cut pUC19 (10 uM in yellow cap labeled "TailVF Fwd") 2.5 uL Reverse primer preceding the chromophore (10 uM in yellow cap labeled "CPUni Rev") 2.5 uL 2x Phusion High-Fidelity PCR Master Mix 25 uL -
Prepare the reaction in a PCR tube to produce the chromophore mutant library fragments:
To Reaction Volume (50 uL Totral) DNase/RNase-Free Water 18 uL 200 ng miniprepped amilCP (100 ng/ul in blue cap tube) 2 uL Reverse primer with tail that overlaps with cut pUC19 (10 uM in purple cap labeled "TailVR Rev") 2.5 uL Forward primer defining the chromophore library and overlaps with the universal reverse primer (10 uM in purple cap labeled "CPLib Fwd") 2.5 uL 2x Phusion High-Fidelity PCR Master Mix 25 uL -
Thermocycle reactions in a PCR program with with following program.
- Initial heat denature DNA at 98C for 30 seconds
-
Repeat the following for 35 cycles:
- Denature DNA at 98C for 10 seconds
- Anneal primers at 60C for 20 seconds
- Extend from primers with DNA polymerase primers at 72C for 1 minute. (Account for 1kb/30sec processivity)
- Final extension at 72C for 5 minutes.
- Hold at 4C until samples are retrieved.
-
Prepare the reaction in a PCR tube to produce the universal chromophore-preceding fragment:
-
Purify the DNA products from your reactions using the Zymo DNA Clean & Concentrator™-25 Kit. The following protocol is copied from Zymo Research and based on silica adsorption.
- For each reaction: In a 1.5 ml microcentrifuge tube, add 5x volumes of DNA Binding Buffer to each volume of reaction. Mix briefly by vortexing.
- Transfer each mixture into a separate Zymo-Spin™ Column in a Collection Tube.
- Centrifuge for 30 seconds at 13,000 rpm (~17,900 x g). Discard the flow-through.
- Add 200 µl DNA Wash Buffer to the column. Centrifuge at for 30 seconds at 13,000 rpm (~17,900 x g). Repeat the wash step.
- Transfer the column to a new 1.5 ml microcentrifuge tube and add 25 µl of DNase/RNase-free water directly to the column matrix. Let sit at room temperature for one minute.
- Centrifuge for 30 seconds at 13,000 rpm (~17,900 x g) to elute.
-
Measure the concentration of your purified DNA using the Nanodrop spectrophotometer.
- Clean stage with Kimwipe soaked with DI water.
- Wipe stage with dry Kimwipe.
- Add 2 uL DI water on stage and make blank measurement.
- Wipe stage with dry Kimwipe.
- For each DNA sample, add 2 uL and measure and record the concentration (ng/uL). Wipe stage dry with Kimwipe between measurements.
- To finish, clean stage with Kimwipe soaked with DI water and then wipe dry.
-
Setup your Gibson Assembly reaction in PCR tubes.
- In a 10 ul total volume, mix 100 ng of cut vector (PvuII digest) with a 2-fold excess of gene fragment (PCR reactions) in DNase/RNase-free water. Molar ratios can be approximated by the lengths of the DNA products. Note, the large fragment of the digested pUC19 is 2,364 bp. The gene fragment amplicons are 353 bp and 777 bp. (( [ng of vector] x [kb size of insert] ) / ( kb size of vector )) x ( insert:vector ratio) = ng of insert required
- Combine with 10 ul 2x Gibson Master Mix.
- Incubate in the thermocycler at 50C for at least 15 minutes.
-
Heat shock assemblies into chemically competent E coli (stored across the hall in the -80C).
- Move Gibson assembly into a cold block on ice.
- Thaw chemically competent cells on ice for 10 minutes.
- Aliquot 50 ul cells into a PCR strip kept in a cold block on ice.
- Add 5 µl of the assembled product to the competent cells. Mix gently by pipetting up and down or by flicking the tube 4–5 times. Do not vortex.
- Place the mixture on ice for 20 minutes. Do not mix
- Heat shock in the thermocycler at 42C for 30 seconds. Do not mix.
- Transfer tubes to ice for 2 minutes.
- Combine heat-shocked cells with 500 ul of room-temperature SOC media in a new 1.5 ml microcentrifuge tube.
- Spread 250 µl of the cells onto the ampicillin selection plates.
- Incubate overnight at 37C in the warm room.
Pro Challenge
Individually test the PCR performance of a set of shifted forward primers that change the chromophore-encoding sequence and have the template-mismatched positions closer towards the 3' end of the primer. Evaluate by gel electrophoresis.
Discussion Questions
- What are the Gibson overlap sequences in our DNA assembly design? How is an overlap created between the cut pUC19 and the PCR amplified parts of the amilCP gene? See assignment lecture slides
- What is the melting temperature for each overlap sequence and how do they compare to the incubation temperature for the Gibson assembly reaction? See http://biotools.nubic.northwestern.edu/OligoCalc.html
- What is the purpose of each enzyme in the Gibson assembly mix?
Bonus Questions
- Our 50 ul PCR reactions included 0.5 uM of both forward and reverse primer and 200 ng of template DNA in the form of a 2,924 bp plasmid. Assuming perfect doubling of DNA copies with each PCR cycle, how many cycles does it take to use all primers? Recall, primers are extended to form copied products. See https://nebiocalculator.neb.com
- After the heat shock step in our experiment, how can we select for bacteria that contain only the original mUAV plasmid?
- Read the following post and its references: https://bitesizebio.com/2267/plasmid-retention/ Comment on the likelihood of a colony containing two plasmids from our heat shock into NEB 10-beta cells.