Seminar 1: Materials
A design and manufacturing perspective on materials. Why I would choose which material.
Materials.
A designer needs to specify materials along the designing process. The key factors that will determine the use of specific materials are:
Physical properties
Physical properties of materials will allow us to distinguish which material is a better candidate for certain applications.
https://www.youtube.com/watch?v=K4WQMQiAOAU
Stiffness,
Stiffness is the extent to which an object resists deformation in response to an applied force.
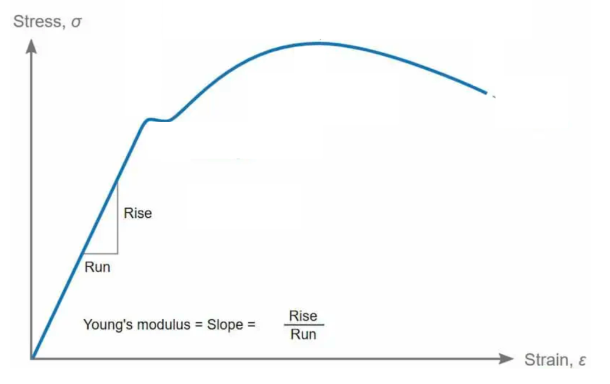
Strength, (US, YS)
Defined as the ability to withstand an applied load without failure or plastic deformation.
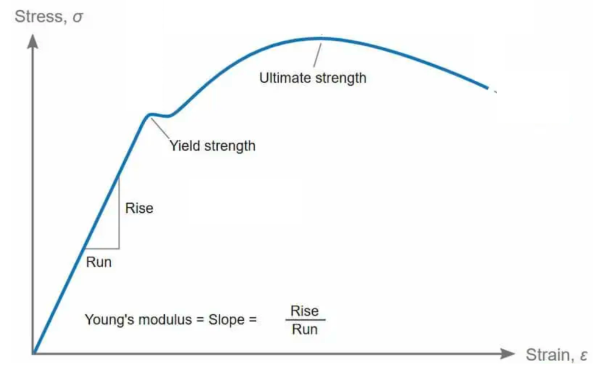
Ductility and Toughness
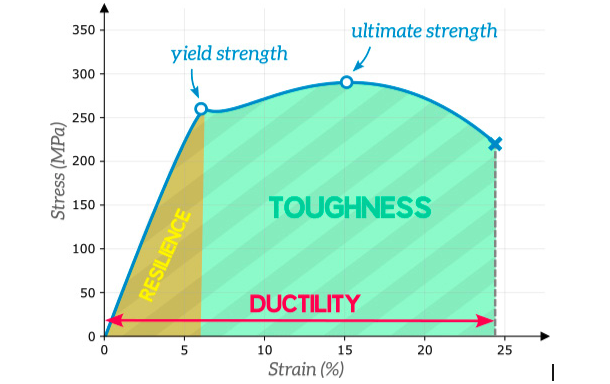
"https://efficientengineer.com/material-strength-ductility-toughness/"
Ductility is the ability of a material to absorb energy. In other words, it is the ability of a material to keep deforming plastically before fully fracturing.
Toughness is the amount of energy per unit volume that a material can absorb before rupturing
This is a fun paper that talks about the almost exclusive relationship between strength and toughness "https://www.nature.com/articles/nmat3115"
All this is at room temperature!
Temperature changes mechanical properties, and our machines could be thought to operate at non-room temperature. Take this into account or:

Engineering families of materials and properties quantification.
Ductile vs. brittle materials
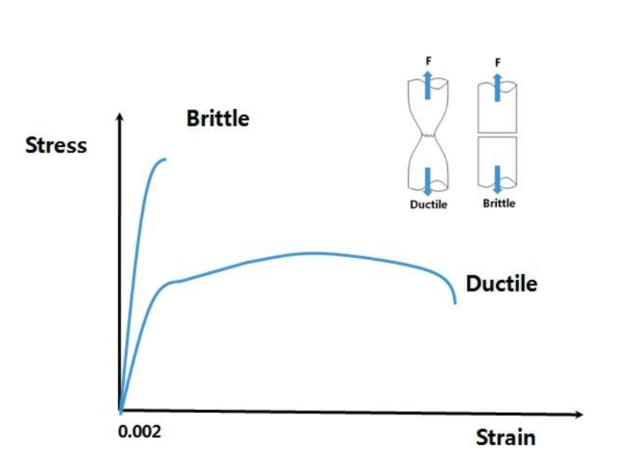
Ductile materials tend to perform equally in compression and tension modes
Brittle materials really like working in compression
The concept of yield strength doesn't make sense here
Big scope material families good candidates for this class and why (properties)
Metals
- Availability,
- Alloys!
Essential metals are: Iron, Aluminum, Titanium, Copper, Nickel, Magnesium.
What makes metals have plastic deformation…?Metals show a crystalline structure, arranged in atomic lattices (FCC, BCC, HCP), but in the real world, the atomic lattice is not perfect and it presents defects. Some of those defects are call dislocations. The high-density packing of the metals and its dislocations is what makes metal behave the way it does.
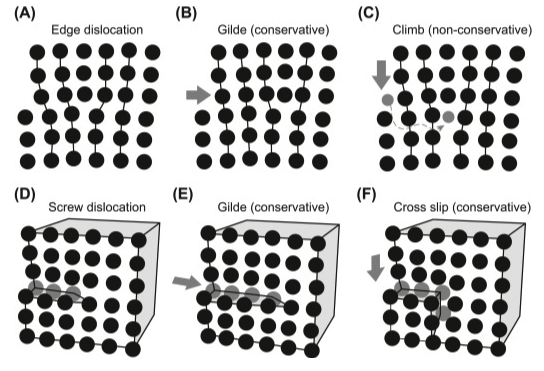
https://www.sciencedirect.com/topics/engineering/dislocation-motion
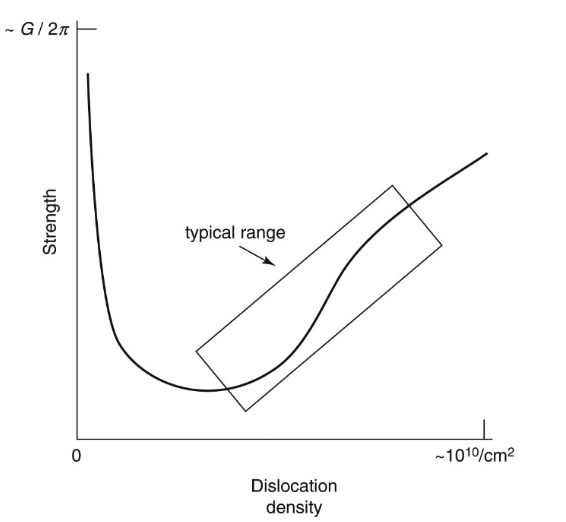
The intuition for this is like the compaction phase of a foam.
Manipulating metals changes their mechanical properties
-heating-cooling hardens metals as it changes their composition
- growing oxides (oxidation or anodization) can make the alloy have a better life; oxides tend to be harder than the base material, offering a layer of protection. This layer also helps with oxidation.
Ceramics
-Do not exhibit plastic deformation. The reason is the nature of their atomic chemical bondings (covalent and ionic). Their delta of energy to slip is much higher than metals, making them reach fatal loads way before it could possibly slip atomically.
Polymers
- Cheap
- Tend to be Amorphous, but long chains can orient and then we call them crystalline
- polymers
- Little corrosion!
- Chemically resistant
- Good in tension and compression
- toughness
Composites
-Super low density vs. stiffness and strength
-(almost) no fatigue
- complex shape without subtractive processes
- electrical resistivity
- great at thermal shocks, no brittleness when cooled
- custom anisotropic mechanical properties; we align the fibers in the load's directions
Ashby Charts:
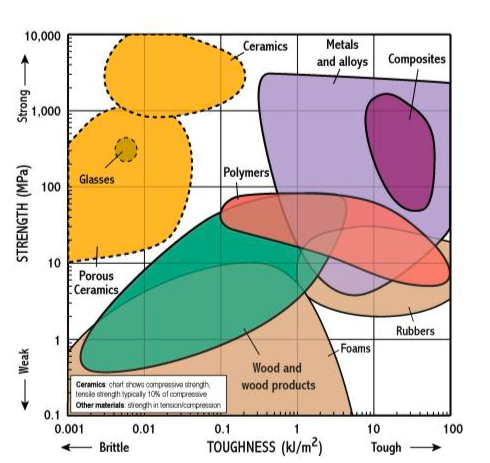
There is a need to compare mechanical properties visually in the early process of an engineering design process.
Michael Farries Ashby , became famous (and with a reason) by developing a visual path to do this with the famous Ashby plots. These are some generalistic comparisons of material families showing any desired mechanical property relationship:
This webpage is fun: http://www-materials.eng.cam.ac.uk/mpsite/interactive_charts/
Economic of Stiffness and Strength
At the end of the day, we have constrained budgets that don't allow us to pick the best but instead, we pick whatever is good enough.
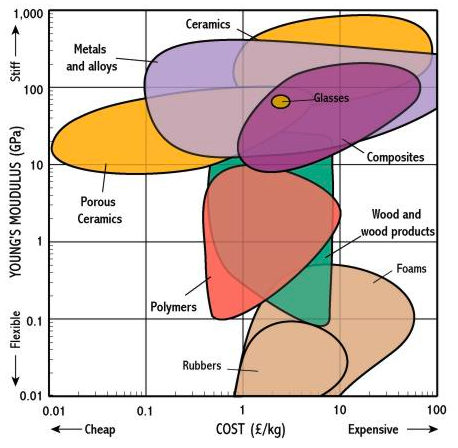
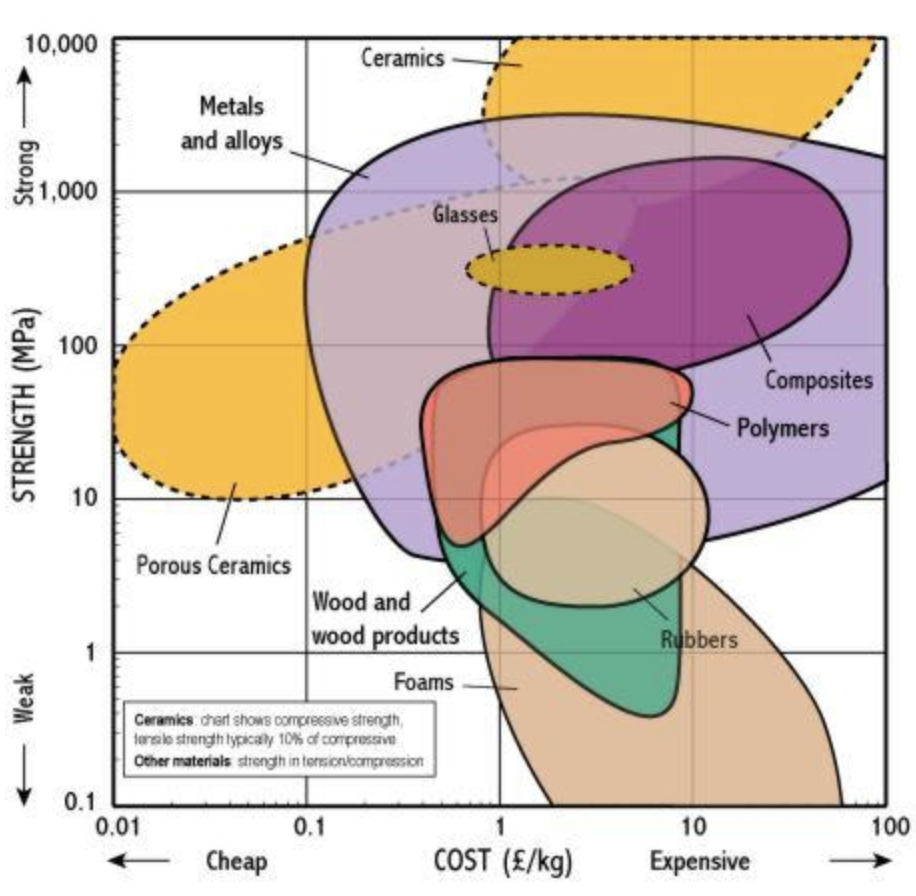
Neil did for a metric of stiffness vs. volume metric for some easy-to-obtain materials:
https://gitlab.cba.mit.edu/neilg/urumbu/-/tree/master/components?ref_type=heads© 2024 MIT 865. All Rights Reserved.